MELBOURNE — Tired of hearing about deadly infections? Here’s some good news. Researchers have created a novel superbug-killing coating that may be applied to wound dressings, surgical instruments, and implants to protect the body from potentially fatal microbial infections. The antimicrobial coating is among the thinnest ever produced, and it works against a wide spectrum of drug-resistant microorganisms, including bacterial and fungal cells, without damaging human cells.
Antibiotic resistance is a serious public health concern that leads to the deaths of more than 700,000 people per year. Without new antimicrobial treatments, the number of deaths might climb to 10 million people per year by 2050, costing the U.S. $100 trillion in medical services. Although the health impact of fungal infections is unrecognized, they kill around 1.5 million people worldwide each year, and the number is rising. The common fungus Aspergillus, which may cause fatal secondary infections, is an increasing danger to COVID-19 patients in hospitals.
The unique coating, developed by a research group at RMIT University, is constructed on a super-thin 2D material that, up to this point, was only considered for use in developing new electronics. The main substance, black phosphorus (BP), has been shown to have antibacterial and antifungal effects in previous studies. It has never been thoroughly investigated for therapeutic application, however.
BP is efficient in killing bacteria when placed in nanothin films on materials like titanium and cotton, which are used to manufacture implants and wound dressings.
Dr. Aaron Elbourne, a co-lead researcher, describes the discovery of a single substance that might prevent both fungal infections and bacterial infections as a big step forward. “These pathogens are responsible for massive health burdens and as drug resistance continues to grow, our ability to treat these infections becomes increasingly difficult,” says Elbourne, who is also a postdoctoral fellow in the School of Science at RMIT, in a statement.
“We need smart new weapons for the war on superbugs, which don’t contribute to the problem of antimicrobial resistance. Our nanothin coating is a dual bug killer that works by tearing bacteria and fungal cells apart, something microbes will struggle to adapt to. It would take millions of years to naturally evolve new defenses to such a lethal physical attack,” adds Elbourne. “While we need further research to be able to apply this technology in clinical settings, it’s an exciting new direction in the search for more effective ways to tackle this serious health challenge.”
Associate Professor Sumeet Walia of RMIT’s School of Engineering, who is also the co-lead researcher, has recently pioneered breakthrough work utilizing BP for technology associated with artificial intelligence and electronics that mimic brain functions.
“BP breaks down in the presence of oxygen, which is normally a huge problem for electronics and something we had to overcome with painstaking precision engineering to develop our technologies,” Walia explains. “But it turns out materials that degrade easily with oxygen can be ideal for killing microbes – it’s exactly what the scientists working on antimicrobial technologies were looking for. So our problem was their solution.”
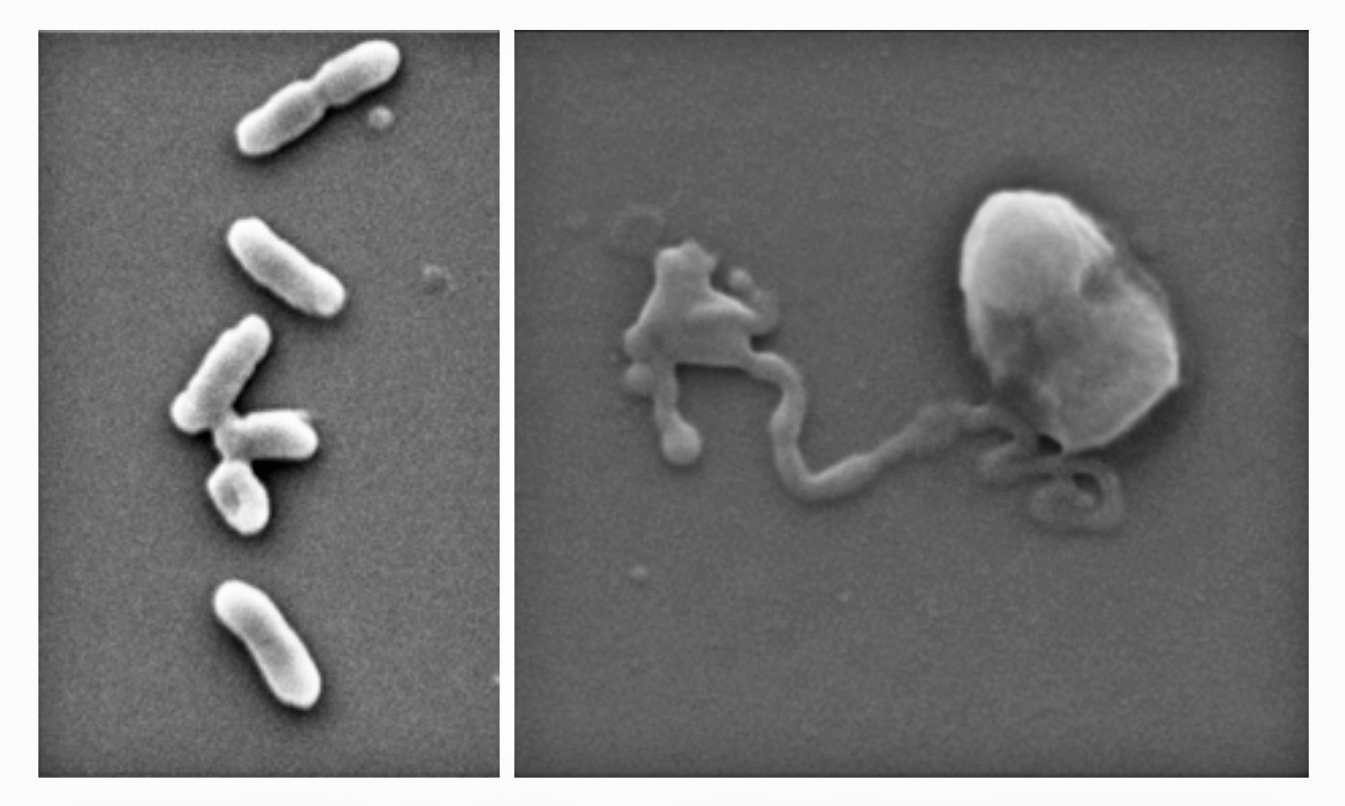
The BP coating works by oxidizing the bacteria and fungal cells’ exterior as it degrades. Cells are ripped apart during this process known as cellular oxidization. The efficacy of nanothin coatings of BP was evaluated against five main bacteria strains, including drug-resistant MRSA and E. coli, as well as five forms of fungi, including Candida auris.
Study co-author Zo Shaw reports that up to 99% of the bacterial and fungal cells were killed in under two hours. Moreover, the BP proceeded to decay during that period and was completely dissolved in 24 hours, indicating that the substance would not collect in the body. The optimal amounts of BP that have a lethal antibacterial action while keeping human cells unharmed and intact were discovered in this study.
The researchers will now investigate other compositions to see how effective they are on a variety of surfaces necessary in the medical field. The team is eager to work with prospective industry associations to further fund the research and help continue the development, which has been granted a temporary patent.
This study is published in the American Chemical Society’s journal Applied Materials & Interfaces.