CHICAGO, Ill. — Doctors usually turn to antibiotics to treat and do away with common bacterial infections. Now, however, a fascinating new study reveals they may also be helpful in battling diseases such as cancer. To be clear, the findings are purely theoretical at this point.
A series of lab experiments, researchers from the University of Illinois-Chicago discovered that they can modify eukaryotic ribosomes to respond to antibiotics in the same way that prokaryotic ribosomes do.
For clarity’s sake, fungi, plants, animals, and humans are all eukaryotes. Why? All of those groups consist of cells that have a clearly defined nucleus. Bacteria, on the other hand, are prokaryotes, which means their cells do not have a definitive nucleus or have one with a different structures, sizes, and properties.
Ribosomes, which are responsible for the protein synthesis inside the cell body, also differ between eukaryotes and prokaryotes.
“Some antibiotics, used for treating bacterial infections, work in an interesting way. They bind to the ribosome of bacterial cells and very selectively inhibit protein synthesis. Some proteins are allowed to be made, but others are not,” says study author Alexander Mankin, the Alexander Neyfakh Professor of Medicinal Chemistry and Pharmacognosy at the UIC College of Pharmacy, in a university release. “Without these proteins being made, bacteria die.”
Teaching the cells some ‘cool tricks’
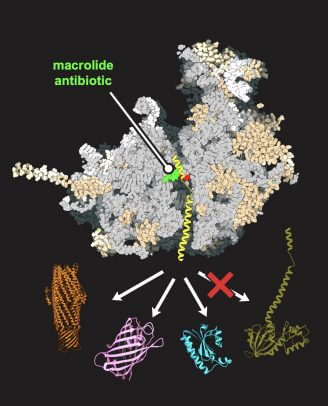
When physicians use antibiotics to treat an infection, the drugs don’t actively change a patient’s cells. This is because antibiotics don’t normally have the means to bind to the differently shaped ribosomes of eukaryotic cells.
“Because there are many human diseases caused by the expression of unwanted proteins — this is common in many types of cancer or neurodegenerative diseases, for example — we wanted to know if it would be possible to use an antibiotic to stop a human cell from making the unwanted proteins, and only the unwanted proteins,” Mankin explains.
In pursuit of some answers, the research team analyzed yeast, a eukaryote featuring cells very similar to human cells. Moreover, to make the process a bit easier, Mankin add they also performed a “cool trick.”
“We engineered the yeast ribosome to be more bacteria-like,” the professor reports.
The team then employed a combination of biochemistry and fine genetics to make changes to just one nucleotide among more than 7,000 in yeast ribosomal RNA. That change was enough to make a macrolide antibiotic (a common variety of antibiotics) “act” on the yeast ribosome.
Understanding the good and bad of human biology
Using that yeast model, researchers applied genomic profiling and high-resolution structural analysis to better understand protein synthesis in the cells. It also allowed them to see how the macrolide interacts with the yeast ribosome.
“Through this analysis, we understood that depending on a protein’s specific genetic signature — the presence of a ‘good’ or ‘bad’ sequence — the macrolide can stop its production on the eukaryotic ribosome or not,” Mankin comments. “This showed us, conceptually, that antibiotics can be used to selectively inhibit protein synthesis in human cells and used to treat human disorders caused by ‘bad’ proteins.”
“Now that we know the concepts work, we can look for antibiotics that are capable of binding in the unmodified eukaryotic ribosomes and optimize them to inhibit only those proteins that are bad for a human,” he concludes.
The study appears in the journal Nature Communications.