BALTIMORE — Chlorine is the most common way to sterilize water in the U.S., but it’s also a powerful substance that is toxic in high doses. And when it comes in contact with other elements in water, chlorine can produce toxic byproducts, a new study shows.
Scientists at Johns Hopkins University teamed up with researchers from Switzerland and the University of California, Berkeley to learn more about the impact of chlorine on water toxicity levels. Their discovery of obscure, but toxic byproducts in drinking water made them question whether we are using too much chlorine and whether there are other viable options to disinfect water.
“There’s no doubt that chlorine is beneficial; chlorination has saved millions of lives worldwide from diseases such as typhoid and cholera since its arrival in the early 20th century,” says lead study author Carsten Prasse, an assistant professor of Environmental Health and Engineering at Hopkins, in a statement.
“But that process of killing potentially fatal bacteria and viruses comes with unintended consequences,” he adds. “The discovery of these previously unknown, highly toxic byproducts, raises the question how much chlorination is really necessary.”
Prasse explains that phenols were an important part of their study. Phenols are chemical compounds used in many personal care products and pharmaceuticals. They are perfectly natural, but when these products enter the water supply, they become part of the mix coming into contact with chlorine. And phenol plus chlorine equals strange new chemical combinations, some of which are harmful to our health.
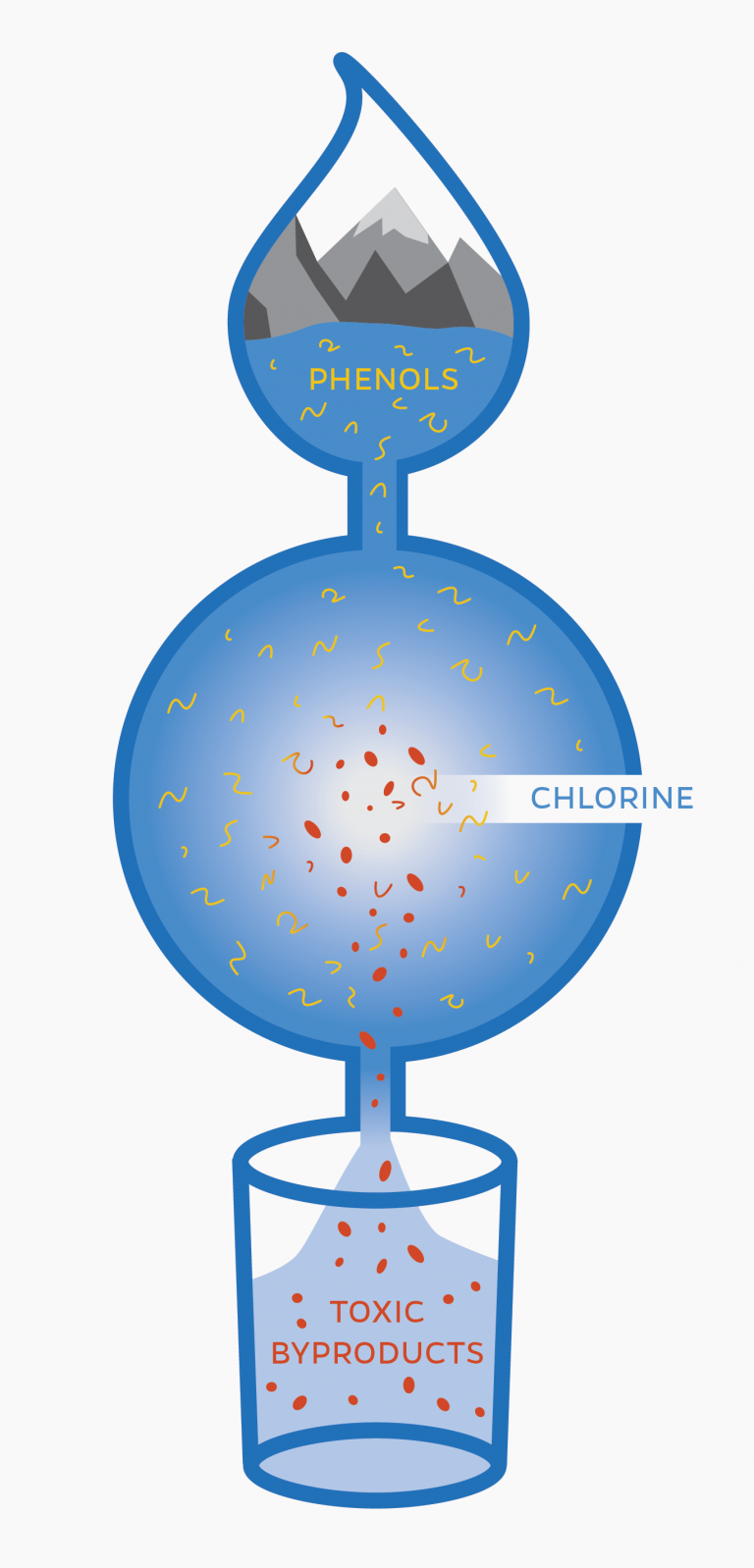
Previous research has linked electrophiles, a type of chemical compound, to a variety of diseases. For this study, Prasse and the other researchers used a toxicology method to find out how compounds react with such biological molecules as proteins and DNA.
Prasse and his colleagues first disinfected water with chlorine just as most municipalities would do: with an extra dose of chlorine to remove offensive but harmless compounds that consumers do not want to taste or smell.
They then performed the same tests, but this time added to the water an amino acid (N-α-acetyl-lysine) that mimics the lysine that is the basis of many human proteins. They left the concoction alone for a day, then used mass spectrometry to check the chemicals in the mix and to find out how the electrophiles had reacted with the added amino acid.
The experiment produced some not-so-tasty compounds, including one known as BDA, a highly toxic compound and known carcinogen. Researchers say this study is the first to reveal the presence of BDA in chlorinated water.
Prasse cautions that these results are from a lab-based study and may not reflect what is found in a typical glass of drinking water. But after finding novel toxic byproducts in their laboratory experiments, study authors hope to raise awareness about other options available to disinfect drinking water. Among these are the use of UV treatments, ozone or simple filtration methods.
“In other countries, especially in Europe, chlorination is not used as frequently, and the water is still safe from waterborne illnesses,” states Prasse. “In my opinion, we need to evaluate when chlorination is really necessary for the protection of human health and when alternative approaches might be better.”
Prasse says the study also shows the need for new ways to analyze just what is in our water supplies. “One reason regulators and utilities are not monitoring these compounds is that they don’t have the tools to find them,” he concludes.
The study is published in the journal Environmental Science & Technology.
