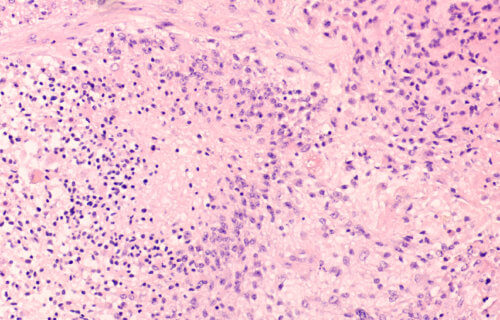
FRANKFURT, Germany — Glioblastoma is the most common and deadliest form of brain cancer in humans. Finding a way to destroy these tumors could save countless lives from a disease which has one of the lowest survival rates. Now, researchers in Germany say a common drug for diarrhea can break up these cancer cells. Their study reveals loperamide can effectively kill glioblastoma tumors by causing the cancerous cells to eat themselves alive.
The team from Goethe University is led by Dr. Sjoerd van Wijk from the Institute of Experimental Cancer Research in Pediatrics. Van Wijk discovered loperamide’s positive cancer-fighting effects two years ago, but didn’t know how the drug did this until now.
Making cancer recycle itself into non-existence
Researchers say when you add loperamide to certain cancer cells it creates a stress response in the endoplasmic reticulum (ER). The ER is a cell substructure that controls key steps in protein synthesis. When the ER comes under stress, it starts to degrade and the cell eventually self-destructs.
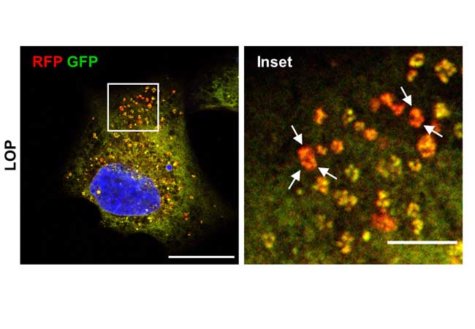
The process, called autophagy-dependent cell death, takes place when cells undergo hyperactivated autophagy. The normal autophagy process helps the cells break down and recycle valuable parts of damaged or unnecessary cell structures. This helps cells to survive even when suffering from a nutrient deficiency.
When scientists trigger hyperactivated autophagy, the cancerous tumor cells recycle so much of the cell’s material they become unstable and can’t survive anymore.
“Our experiments with cell lines show that autophagy could support the treatment of glioblastoma brain tumors,” says van Wijk in a university release.
The average survival length for both adults and children with glioblastoma is about 18 months. Less than five percent of patients live longer than five years after their diagnosis. The German team notes that even chemotherapy has a poor track record of treating this brain cancer.
The gene that kills glioblastoma cells
The new study reveals that the gene “Activating Transcription Factor” (ATF4) plays an important role in causing ER stress. Researchers discovered that ATF4 is present in large amount both during ER stress and when cells are exposed to loperamide. The team concludes this gene helps to trigger the destruction of the ER membrane (reticulophagy).
“Conversely, if we block ATF4, far fewer cells in a tumor cell culture die after adding loperamide,” van Wijk explains.
Study authors add that they could see ER debris inside the loperamide-treated cells when viewing them through an electron microscope.
“ER degradation, that is, reticulophagy, visibly contributes to the demise of glioblastoma cells.”
Luckily, the study also reveals that loperamide only triggers hyperactivated autophagy in cancer cells. Other cells in the body do not break down when exposed to the anti-diarrhea medication.
“Normally, loperamide, when taken as a remedy against diarrhea, binds to particular binding sites in the intestine and is not taken up by the bowel and is therefore harmless,” the researchers explain.
Finding a way to get loperamide to the brain
The German team says this breakthrough is a long way from a finished cancer treatment. Van Wijk notes that scientists need to find a way for loperamide to cross the blood-brain barrier so it can attack brain tumors inside a living patient. Previous studies have already suggested that nanoparticles may be able to achieve this. In the meantime, the study authors also believe their findings reveal how other diseases can be treated by focusing on ER degradation.
“Our findings also open up exciting new possibilities for the treatment of other diseases where ER degradation is disrupted, such as neurological disorders or dementia as well as other types of tumor,” van Wijk concludes.
The study appears in the journal Autophagy.