LEUVEN, Belgium — Although children with Down syndrome can lead a healthy and normal life, the genetic disorder is synonymous with facial changes which stay with these individuals permanently. A new study finds there may be a way to lessen the severity of these changes, thanks to the chemicals in green tea. Researchers from Belgium and Spain say low doses of green tea extracts can reduce facial dysmorphology in children with Down syndrome.
While the news is exciting, scientists note that early intervention is key. Their study finds children receiving green tea extracts during the first three years of life saw a significant reduction in facial changes due to Down syndrome. However, the same results did not appear in the older children receiving the supplements.
A low dose also appears to be important in Down syndrome dysmorphology. Researchers add that experiments on mice reveal high doses of the extract can actually disrupt facial and bone development even further.
Green tea cancer killer may also help with Down syndrome
Researchers say the presence of a third copy of chromosome 21 causes the genetic disorder. This extra copy triggers an overexpression of genes that results in both physical and intellectual disabilities. One of these genes, DYRK1A, is responsible for altering the brain and bone development in Down syndrome patients.
Green tea contains a compound called EGCG, or epigallocatechin-3-gallate. Previous studies have discovered that this natural antioxidant can help boost proteins which fight cancer. Researchers say EGCG also has the ability to inhibit DYRK1A. They add that the green tea extract may boost cognitive function in young adults with Down syndrome too.
A team from the Centre for Genomic Regulation (CRG), European Molecular Biology Laboratory (EMBL), University of Barcelona in Spain, and KU Leuven in Belgium conducted two studies on the green tea supplement’s impact on Down syndrome patients.
The first involved experiments at KU Leuven, where study authors gave mice either low or high doses of green tea extracts in their drinking water. These treatments began while the mice pups were still in their mother’s womb. The second observational study involved children with and without Down syndrome.
Green tea’s ‘positive effect’ on Down syndrome patients
“The low dose treatment had a positive effect on mice that are a model of Down syndrome,” co-lead author Professor Greetje Vande Velde from KU Leuven says in a university release. “Sixty percent of them showed a facial shape similar to the control group.”
“The high dose treatment, however, generated very mixed results, and even disrupted normal facial development in some cases, causing additional dysmorphology. This occurred in all mice, in the model of Down syndrome as well as in the control group,” Vande Velde adds.
In the observational study, 287 children between zero and 18 years-old received EGCG supplements. Researchers note the group self-medicated and did not have to follow a specific protocol for taking the green tea extract.
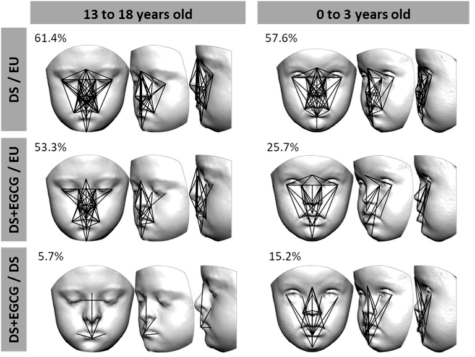
“All participants were photographed from various angles to create a 3D model of their faces,” explains Neus Martínez-Abadías from the University of Barcelona.
“We use 21 facial landmarks, and the distances between them, to compare the faces of the participants. In the youngest group between 0 and 3 years, we observed that 57 percent of the linear distances are significantly different when you compare the faces of children with Down syndrome that never received the treatment to those of children that do not have Down syndrome. For babies and toddlers who did receive EGCG treatment, this difference was much smaller, only 25 percent. After green tea supplementation, the facial dysmorphology decreases and the children with or without Down syndrome look more alike.”
Early treatments may be the only solution
Study authors note that treatments on adolescents did not produce the same results younger children experienced. They add that more research will help advise parents who may look to purchase these commercially-available supplements on their own.
“Even when treated with green tea extracts, children with Down syndrome still show a difference of more than 50 percent compared to the control group. These findings suggest that the green tea supplements only affect facial development when they are administered in the early stages of life when the face and skull are rapidly growing,” Prof. Martínez-Abadías reports.
“Despite the potential benefits we observed, we must handle these findings with caution considering they are preliminary and based on an observational study,” Prof. Vande Velde cautions. “Much more research is necessary to evaluate the effects of EGCG-containing supplements, the appropriate dose and their therapeutic potential in general. We also need to take into the account possible effects on other organs and systems, not just on the development of the face. This requires first more basic research in the lab with mice, and then clinical studies with more participants and controlled use of these supplements.”
“Our findings suggest that effects of EGCG strongly depend on the dose,” Martínez-Abadías concludes. “EGCG products are commercially available and they are used regularly as a general health-promoting compounds. However, it’s important to follow the European Food Safety Authority recommendations regarding the maximal intake and to always consult a physician before taking the supplements. Our research shows potential beneficial effects of facial development at low doses, but at very high doses they can produce unpredictable effects in mice. More research is needed in humans to determine the optimal dose at each age that maximizes the potential benefits.”
The study appears in the journal Scientific Reports.
