NEW YORK — Changes in the brain during the first month of a baby’s development in the womb may lead to schizophrenia later in life, a new study reveals. Researchers from Weill Cornell Medical College have discovered that a drop in the expression of two genes causes significant brain cell changes that contribute to the disease. Interestingly, the team created their own “mini-brains” to make this discovery and find out what’s going on inside the developing mind.
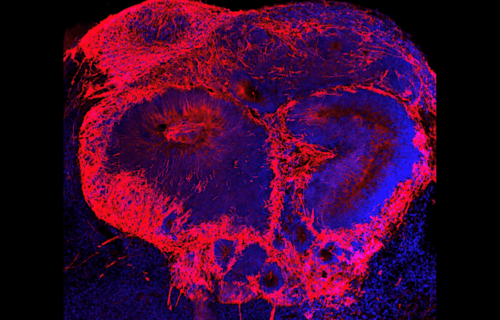
Researchers used stem cells from patients with schizophrenia and from people without any brain disease to grow 3D organoids (“mini-brains”) in a lab setting. From there, they were able to compare these organoids and see the differences in how the stem cells grew into working brain cells.
Results show that mini-brains from schizophrenia patients experience reduced expression from two genes, which disrupts early brain development and also causes more brain cell death. Specifically, scientists found that schizophrenia patient samples saw a drop in the expression of a regulator of gene expression called BRN2 and a cell growth promoter called pleiotrophin.
“This discovery fills an important gap in scientists’ understanding of schizophrenia,” says senior author Dr. Dilek Colak, an assistant professor of neuroscience at Weill Cornell Medicine, in a university release. “There were hints schizophrenia started during early development, but we didn’t have proof.”
When does schizophrenia really begin?
Typically, schizophrenia symptoms appear during adulthood. However, previous studies of schizophrenia patients after death have revealed that their brains had large cavities (ventricles) and other differences which scientists believe formed earlier in life.
By growing their own mini-brains, the Weill Cornell team was able to examine the possible origins of the disease as the brains grew from stem cells. They used single-cell RNA sequencing to compare the gene expression of 21 organoids, grown from donor samples of people with and without the disease.
“We found a common pathology among all the patients with schizophrenia despite each of the patients having distinct disease presentations,” Dr. Colak reports.
The team also found that replacing the missing BRN2 restored brain cell function in the organoids. Adding more pleiotrophin helped to reduce levels of brain cell death. Researchers add that if future studies confirm this link, it could lead to a new gene therapy to correct brain cell abnormalities.
“We’ve made a fundamental discovery providing what we think is the first evidence in human tissue that multiple cell-specific mechanisms exist and likely contribute to risk of schizophrenia,” says first author Dr. Michael Notaras. “This forces us, as a field, to reconsider when disease truly begins and how we should think about developing the next generation of schizophrenia therapeutics.”
Certain cell changes during pregnancy may play a role
Study authors also used their mini-brains to examine the role of specific cell types which may play a role in the development of schizophrenia. They focused on endothelial cells, which line the blood vessels and release immune molecules called cytokines. Organoids from schizophrenia patients had excess amounts of endothelial cells during early development stages, which can cause an overactive inflammatory response to infections.
“This may explain the link between maternal infections during pregnancy and schizophrenia seen in mouse studies,” Dr. Colak concludes. “The technique could be used to study the early life pathology of late-onset neuropsychiatric or neurodegenerative diseases like Alzheimer’s disease or Huntington disease.”
The study is published in the journal Molecular Psychiatry.