CHAPEL HILL, N.C. — A signaling pathway in the body that helps fight off infections may also hold the key to a breakthrough in breast cancer research. Researchers from the University of North Carolina say activating this path can allow genetically engineered cells destroy breast cancer tumors in mice.
Their study finds this immunotherapy activates the “STING” pathway, an immune cell signaling system which normally triggers inflammation in the body when a virus or bacteria invades. The treatment boosts the ability of T-cells to eradicate the disease.
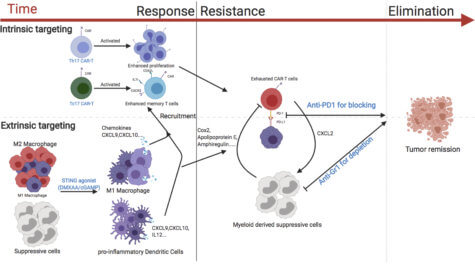
These cancer fighters are called chimeric antigen receptor (CAR) T-cells and researchers use them to treat blood cancers in humans. They may also be successful against solid tumors if combined with other immunotherapeutic approaches.
CAR T-cells are a type of white blood cell that have been genetically modified to attack cancer cells with specific proteins on their surfaces. They have also been used to treat lymphomas, tumors that begin in the glands. Ongoing clinical trials are currently testing their abilities to fight many other types of blood cancer.
“However, the clinical activity of CAR T cells in patients or animal models with solid tumors has been modest,” says lead author Professor Jonathan Serody in a media release.
Cancer’s roadblocks to defeating tumors
Study authors say these cells may be less effective at attacking solid tumors because they have to migrate into the tumors and then survive long enough to kill all of the cells. Moreover, the cells and molecules surrounding them are often “immuno-suppressive” and activate a “checkpoint” that stops T-cells.
In a mouse model of breast cancer, the researchers treated the animals with a drug called cGAMP. This fired up the STING pathway and created a pro-inflammatory environment within the tumors. Researchers say this improved the ability of the CAR T-cells to gather and attack cancer cells.
Joining forces to eliminate breast cancer
The accumulation increased when scientists infused the mice with T-cells that produce the immune signaling molecule IL-17A. Prof. Serody notes the attack on cancer cells could be sustained for longer periods if the mice also receive therapeutic antibodies. This combination depleted immuno-suppressive cells and prevented the checkpoint from deactivating CAR T-cells. The study reveals that combining all of these approaches leads to the complete annihilation of breast cancer tumors.
“cGAMP is in clinical trials for the treatment of patients with cancer,” Prof Serody reports. “There are multiple ongoing clinical trials using approaches to inhibit immunosuppressive cells for patients with malignant disease, and there are clinical trials currently evaluating the combination of CAR T cells with immune checkpoint blockade.”
“Together therefore, our data suggest a viable strategy for boosting CAR T activity in solid tumors,” he concludes.
Researchers hail immunotherapy as a new era in cancer treatment, priming the body’s own immune cells to fight the disease.
According to the Centers for Disease Control and Prevention, cancer is the second leading cause of death in the United States, behind only heart disease. About 250,000 women are diagnosed with breast cancer each year.
The study appears in the Journal of Experimental Medicine.
SWNS writer Mark Waghorn contributed to this report.
