PITTSBURGH, Pa. — One of the many goals of stem cell technology is to be able to grow an organ in a laboratory to replace an organ that is diseased, damaged or failing. One major milestone in the development of this technology at the University of Pittsburgh is receiving acclaim. Incredibly, scientists have grown a miniature liver out of human skin cells, transplanted it into rats, and allowed the liver to function in the animal for four days.
These livers were created as a proof-of-concept for stem cell organ growth.
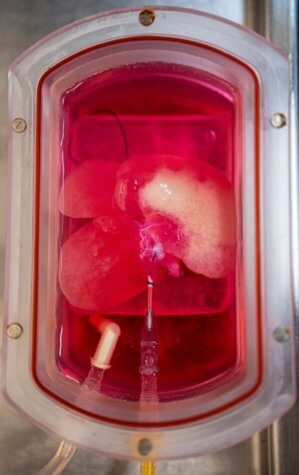
The mini livers the scientists created function just like real ones. They secrete bile acids and urea to digest and process food. Also, these livers are produced relatively quickly. A liver normally takes about two years to fully mature — these livers can be grown in less than a month.
“Seeing that little human organ there inside the animal – brown, looking like a liver – that was pretty cool. This thing that looks like a liver and functions like a liver came from somebody’s skin cells,” says senior author Dr. Alejandro Soto-Gutierrez, an associate professor of pathology at Pitt and faculty member of both the McGowan Institute for Regenerative Medicine and the Pittsburgh Liver Research Center, in a media release.
The researchers had to reengineer the human skin cells so they could properly function as a liver. First they reprogrammed the skin cells into stem cells. The stem cells were coaxed into various types of cells found in the human liver. Researchers removed cells from rat livers so it could serve as a “scaffolding” for the stem cells they created. The human liver cells were seeded into this scaffolding, and finally the livers were transplanted into the rats.
The team only transplanted livers into five rats, but they write that all five of the livers were functioning as expected. Four days after the livers were transplanted in the rats, they found human liver proteins in the blood serum of the rats.
The authors note that after four days there were some unwanted side-effects to the surgery — there were problems with blood flow around the site of the graft (where the liver was transplanted). They say that some of the next steps in the creation of these livers is to improve safety issues and create them so they can survive for the long-term.
“The long-term goal is to create organs that can replace organ donation, but in the near future, I see this as a bridge to transplant,” says Soto-Gutierrez. “For instance, in acute liver failure, you might just need hepatic boost for a while instead of a whole new liver.”
The study is published in Cell Reports.
Like studies? Follow us on Facebook!