DURHAM, N.C. — Urinary tract infections (UTIs) affect 50% of women globally, with some experiencing chronic UTIs at a constant rate. Antibiotics are typically prescribed for sufferers, however, a new study suggests a vaccine may be the key. Researchers say the vaccine could help the body fend off bacteria that cause UTIs entirely.
“Although several vaccines against UTIs have been investigated in clinical trials, they have so far had limited success. There are currently no effective UTI vaccines available for use in the U.S. in spite of the high prevalence of bladder infections. Our study describes the potential for a highly effective bladder vaccine that can not only eradicate residual bladder bacteria but also prevent future infections,” says senior author Soman Abraham, Ph.D., a professor of Pathology, Immunology and Molecular Genetics & Microbiology with Duke University’s School of Medicine.
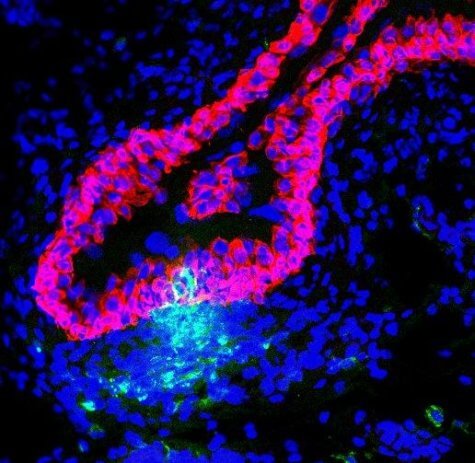
In mouse clinical trials, researchers noticed that the bladder becomes damaged by E. coli bacteria during a UTI. At this point, their immune systems would send repair cells to the damaged area, rather than fighting the bacteria. This allows the bacteria to continue living in the bladder which can stimulate another UTI.
“The new vaccine strategy attempts to ‘teach’ the bladder to more effectively fight off the attacking bacteria. By administering the vaccine directly into the bladder where the residual bacteria harbor, the highly effective vaccine antigen, in combination with an adjuvant known to boost the recruitment of bacterial clearing cells, performed better than traditional intramuscular vaccination,” explains lead author Jianxuan Wu, Ph.D., who recently earned his doctorate from the Department of Immunology at Duke.
According to the test results, the vaccine reprogrammed their immune systems so that immune cells targeted the E. coli bacteria in the bladder. Also, the bacteria in the bladder was completely wiped out. This suggests that the placement of the vaccine directly into the bladder tissue may determine the overall effectiveness of the vaccine.
“We are encouraged by these findings, and since the individual components of the vaccine have previously been shown to be safe for human use, undertaking clinical studies to validate these findings could be done relatively quickly,” said Abraham.
This study is published in Proceedings of the National Academy of Sciences.