CHICAGO, Ill. — When the heart stops beating, the rest of the body begins to shut down. Cells stop receiving oxygen and, eventually, the body dies. However, a recent study finds not every dead cell stays dead forever. Some keep functioning long after death.
Researchers from the University of Illinois-Chicago’s College of Medicine found that specific genes were still active in brain cells — after scientists extracted them from a patient. The team assessed the brain tissue prior to extraction, examining its cellular activity. After extraction, researchers observed cellular activity, as well as the genes related to the activity, at several intervals for a total of 24 hours.
“We decided to run a simulated death experiment by looking at the expression of all human genes, at time points from 0 to 24 hours, from a large block of recently collected brain tissues, which were allowed to sit at room temperature to replicate the postmortem interval,” says Dr. Jeffrey Loeb, the UIC College of Medicine’s head of neurology and rehabilitation, in a university release.
‘Zombie genes’ try to undo death
Loeb and his team discovery that certain genes kept up-regulating (increasing their response to molecular stimulation) after death. These genes — dubbed “zombie genes” — caused neuroglial cells to increase in function.
“Most studies assume that everything in the brain stops when the heart stops beating, but this is not so,” Loeb adds.
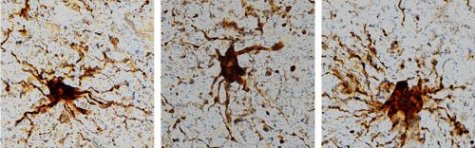
Glial cells, specifically microglia and astrocytes, elicit inflammation as a response to injury or trauma. Microglial cells cause inflammation that astrocytes sustain thereafter. Scientists believe these cells perceive death as a type of injury.
Much like a zombie arm reaching out from the grave, the cells grew long branch-like extensions for up to 24 hours after extraction of the brain tissue. Additionally, compared to their usual sizes, the cells grew to monstrous sizes.
“That glial cells enlarge after death isn’t too surprising given that they are inflammatory and their job is to clean things up after brain injuries like oxygen deprivation or stroke,” Dr. Loeb explains.
Although it isn’t much of a shock to see these particular cells working postmortem, it is an important discovery in the quest to find a cure for diseases such as Alzheimer’s. According to Loeb, many studies involving brain tissue after death do not include cell function or the genes that cause it.
“Our findings will be needed to interpret research on human brain tissues. We just haven’t quantified these changes until now,” the study author continues.
Could zombie genes cure brain disease?
As the head of the University of Illinois NeuroRepository, Loeb and his team have a distinct edge in the matter of brain tissue research. The repository is a reserve for various brain tissue samples including those donated from patients with brain disorders such as Alzheimer’s or schizophrenia, as well as samples collected from normal surgical procedures to prevent epilepsy.
Upon studying these samples, Loeb and his colleagues discovered discrepancies in the regulation of genes in more recent tissue samples when compared to the samples taken after death. They analyzed and compared worldwide samples of recent brain tissue to previous studies regarding gene activity in postmortem samples with or without neurological diseases.
The team discovered that most of the genes (80%) in postmortem tissue were being expressed at their usual rate after about a day. Genes that code for normal, everyday cell activity were among this group. Some genes, such as those responsible for thinking and memory, stopped functioning almost immediately after the patient died. According to Loeb, those genes are especially significant for Alzheimer’s and schizophrenia research.
Timing is everything when working with zombies
Lastly, the team discovered that the “zombie genes” were some of the few genes that actually upregulated themselves, stimulating glial cells to work even faster. These genes exhibited increased expression until 12 hours after death, while the remaining 80 percent of genes had nearly stopped functioning altogether.
“[This] means that researchers need to take into account these genetic and cellular changes, and reduce the post-mortem interval as much as possible to reduce the magnitude of these changes,” Loeb concludes. “The good news from our findings is that we now know which genes and cell types are stable, which degrade, and which increase over time so that results from postmortem brain studies can be better understood.”
This findings appear in the journal Scientific Reports.
