BARCELONA, Spain — Scientists have developed a “living medicine” that has the capability to kill the hospital superbug MRSA. Experiments on mice found it destroyed biofilms of bacteria resistant to antibiotics. The revolutionary treatment could make its way to patients within two years.
“Our technology, based on synthetic biology and live biotherapeutics, has been designed to meet all safety and efficacy standards for application in the lung, with respiratory diseases being one of the first targets. Our next challenge is to address high-scale production and manufacturing, and we expect to start clinical trials in 2023,” says study leader Dr. María Lluch of the Centre for Genomic Regulation (CRG) in a media release.
Remodeling bacteria to go after germs
The novel medicine uses common bacterial cells scientists have genetically-engineered to possess therapeutic properties. The Spanish team removed the bacteria’s ability to cause disease and repurposed it to attack harmful microbes instead. This genetically altered bacteria successfully removed antibiotic-resistant bacteria growing on medical implants.
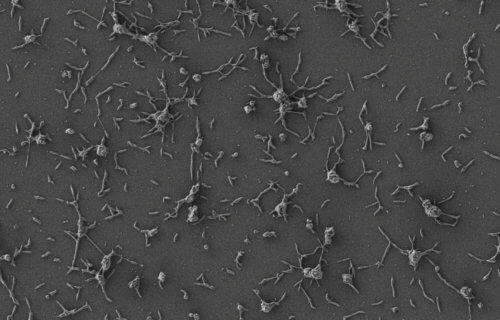
Injecting the therapy under the skin of mice cured infections in 82 percent of the treated animals. It specifically targets biofilms — colonies of bacterial cells that stick together on a surface. These colonies enable ideal growing conditions for forming impenetrable structures that prevent antibiotics or the human immune system from destroying the bacteria embedded within.
Biofilm-associated bacteria can be a thousand times more resistant to antibiotics than their free-floating cousins. MRSA (methicillin-resistant Staphylococcus aureus) is one of the most common types of biofilm-associated bacteria. Infections do not respond to conventional antibiotics, requiring patients to surgically remove any infected medical implants.
Alternative therapies include the use of antibodies or enzymes that are highly toxic for normal tissues and cells, causing undesired side effects. Introducing living organisms that directly produce enzymes in the local vicinity is a safer and cheaper way of treating infections.
Why use bacteria in the first place?
Researchers say bacteria is an ideal vector, as they have small genomes that can be modified using simple genetic manipulation. The team chose to engineer Mycoplasma pneumonia, a common species that lack a cell wall. It made it easier to release the beneficial molecules that fight infection while also assisting it in evading detection from the human immune system.
Other advantages include M. pneumonia’s low risk of developing disease-causing mutations. It also cannot transfer any of its modified genes to other microbes living nearby. M. pneumonia was first modified so it would not cause illness. Further tweaks made it produce two different enzymes. They dissolved biofilms and attacked the cell walls of the bacteria embedded within.
The researchers also changed the bacteria so it secretes antimicrobial enzymes more efficiently. In particular, it treated biofilms building around breathing tubes, as M. pneumonia is naturally adapted to the lung. The modified bacteria may also have long-term applications for other diseases.
“Bacteria are ideal vehicles for ‘living medicine’ because they can carry any given therapeutic protein to treat the source of a disease. One of the great benefits of the technology is that once they reach their destination, bacterial vectors offer continuous and localized production of the therapeutic molecule. Like any vehicle, our bacteria can be modified with different payloads that target different diseases, with potentially more applications in the future,” says study co-author Professor Luis Serrano of CRG.
Living medicines date back centuries to when leeches were first used for bloodletting. Scientists believe they could hold the key to treating a host of diseases, including cancer.
The findings appear in the journal Molecular Systems Biology.
South West News Service writer Mark Waghorn contributed to this report.