(© cristianstorto - stock.adobe.com)
SAN DIEGO, Calif. — A pill used for over two decades to treat the flu may soon find a new job — curing sepsis. Researchers in California say Tamiflu helps fight off the deadly infection by boosting the immune system. The discovery is especially important now, as sepsis is one of the major complications of COVID-19 infections.
Experiments in humans and mice suggest the common prescription medication improves survival chances. Another repurposed drug for preventing secondary heart attacks, the blood thinner Brilinta, also proved just as effective. The study finds these medications stop protective blood cells from dying. They may work better than antibiotics for a condition that the CDC says affects 1.7 million American adults each year.
“In many cases, the antibiotics we give these patients should be able to kill the bacteria, based on lab tests, yet a significant number of patients are not pulling through,” says senior author Professor Victor Nizet from the University of California-San Diego in a release.
“If we can reduce mortality in staph sepsis by 10 or 20 percent by arming or protecting the immune system, we can likely save more lives than discovering an additional new antibiotic that may still not cure the sickest patients.”
What triggers sepsis in patients?
Staph bacteria, or Staphylococcus aureus, causes the bloodstream infection. This antibiotic-resistant germ can be found on the skin of about one in four people right now.
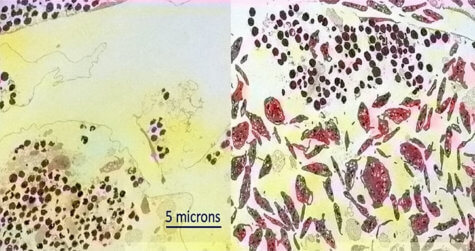
If it enters the body through damaged skin, it can trigger severe illness or death in a third of cases. Last year, researchers discovered sepsis, or blood poisoning, was responsible for one in five deaths globally — more than cancer.
The U.S. Food and Drug Administration approved Tamiflu (which also comes in liquid form) for use in 1999. Brilinta has been available to patients for the last decade. Study authors say they potentially hold the key to combating a health crisis, fueled by resistance to antibiotics.
Sepsis is among the worst problems facing intensive care patients. One in three who die in a hospital have these infections. It occurs when the body responds extremely to an infection, wrecking the immune system. High doses of antibiotics often fail to work.
Blood platelets hold the key to stopping infections
Prof. Nizet and the team discovered a battle occurs between staph bugs and platelets, cells that clot the blood. Levels plummet if the bacteria wins. A study of 49 staph sepsis patients in the U.S. revealed those with fewer platelets are more likely to die.
Study authors collected blood and bacteria samples, along with demographic and health information. Surprisingly, the results show outcomes did not have a link to the immune system’s white blood cell counts. It did however tie into the number of platelets.
Low counts, defined as fewer than 100,000 per cubic millimeter of blood, increased the risk of death. Roughly one-third (31%) died, compared to fewer than six percent of those with platelet counts above that threshold. Tests also showed platelets secrete antimicrobial peptides that help destroy bacteria. At the same time, the staph releases an alpha-toxin that harms platelets.
In addition to poking holes in the blood cells, it convinces them to produce an enzyme that trims off sugar molecules that cover the cell’s surface. Another molecule in the liver, the Ashwell-Morell receptor, recognizes the platelet’s new look and pulls “bald” platelets out of circulation.
Mouse models of the disease identified the two drugs that “tip the balance” back in favor of the patient. Brilinta blocked the alpha-toxin so it can’t injure platelets or stimulate its sugar-removing enzyme. Tamiflu inhibits the latter so the cells don’t go bald and aren’t cleared by the liver — even when the alpha-toxin is around.
Finding new answers in old medications
Either medication maintained more platelets and reduced blood bacteria in the mice. About 60 percent survived their infection, compared to just 20 percent of untreated mice. In humans, side-effects can include nausea, diarrhea, and bleeding. Safety and efficacy trials for patients with staph sepsis would be ideal.
Study authors note that there’s little financial incentive for pharmaceutical companies to develop new treatments with profitable drugs already available. However, repurposing commercially available therapies for sepsis, which can be caused by several other types of bacteria, has many advantages.
“Discovering a new drug is tremendously expensive and takes many, many years,” Prof. Nizet concludes. “But if we look around at what we already have, what we already know to be safe, we may find many opportunities to improve patient outcomes.”
The findings appear in the journal Science Translational Medicine.
SWNS writer Mark Waghorn contributed to this report.







